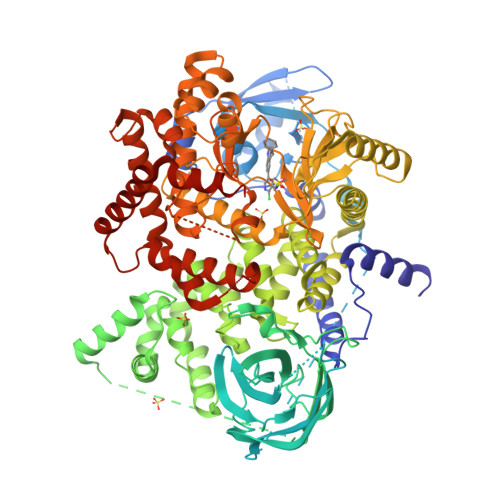
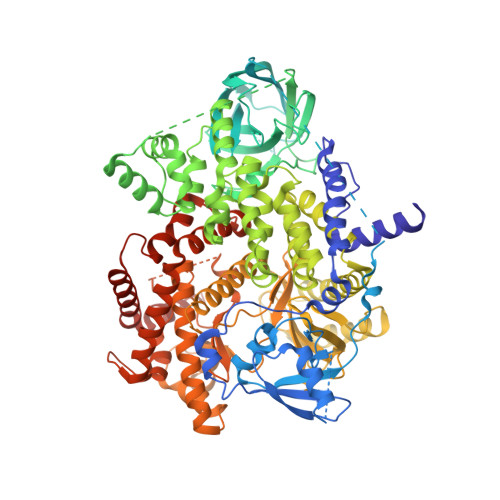
A first-in-class selective inhibitor of EGFR and PI3K offers a single-molecule approach to targeting adaptive resistance.
Whitehead, C.E., Ziemke, E.K., Frankowski-McGregor, C.L., Mumby, R.A., Chung, J., Li, J., Osher, N., Coker, O., Baladandayuthapani, V., Kopetz, S., Sebolt-Leopold, J.S.(2024) Nat Cancer 5: 1250-1266
- PubMed: 38992135
- DOI: https://doi.org/10.1038/s43018-024-00781-6
- Primary Citation of Related Structures:
8SC7, 8SC8, 8SC9 - PubMed Abstract:
Despite tremendous progress in precision oncology, adaptive resistance mechanisms limit the long-term effectiveness of molecularly targeted agents. Here we evaluated the pharmacological profile of MTX-531 that was computationally designed to selectively target two key resistance drivers, epidermal growth factor receptor and phosphatidylinositol 3-OH kinase (PI3K). MTX-531 exhibits low-nanomolar potency against both targets with a high degree of specificity predicted by cocrystal structural analyses. MTX-531 monotherapy uniformly resulted in tumor regressions of squamous head and neck patient-derived xenograft (PDX) models. The combination of MTX-531 with mitogen-activated protein kinase kinase or KRAS-G12C inhibitors led to durable regressions of BRAF-mutant or KRAS-mutant colorectal cancer PDX models, resulting in striking increases in median survival. MTX-531 is exceptionally well tolerated in mice and uniquely does not lead to the hyperglycemia commonly seen with PI3K inhibitors. Here, we show that MTX-531 acts as a weak agonist of peroxisome proliferator-activated receptor-γ, an attribute that likely mitigates hyperglycemia induced by PI3K inhibition. This unique feature of MTX-531 confers a favorable therapeutic index not typically seen with PI3K inhibitors.
Organizational Affiliation:
Department of Radiology, University of Michigan, Ann Arbor, MI, USA.