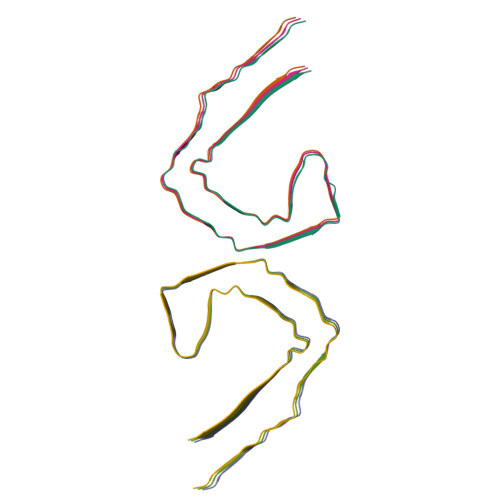
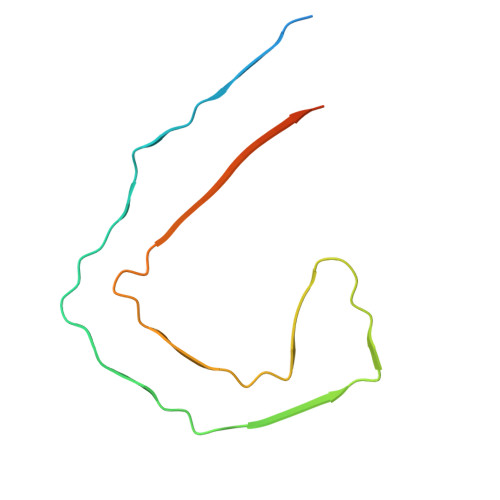
Post-Translational Modifications Control Phase Transitions of Tau.
Powell, W.C., Nahum, M., Pankratz, K., Herlory, M., Greenwood, J., Poliyenko, D., Holland, P., Jing, R., Biggerstaff, L., Stowell, M.H.B., Walczak, M.A.(2024) bioRxiv
- PubMed: 38559065
- DOI: https://doi.org/10.1101/2024.03.08.583040
- Primary Citation of Related Structures:
9HBB - PubMed Abstract:
The self-assembly of Tau(297-391) into filaments, which mirror the structures observed in Alzheimer's disease (AD) brains, raises questions about the role of AD-specific post-translational modifications (PTMs) in the formation of paired helical filaments (PHFs). To investigate this, we developed a synthetic approach to produce Tau(291-391) featuring N-acetyllysine, phosphoserine, phosphotyrosine, and N-glycosylation at positions commonly modified in post-mortem AD brains, thus facilitating the study of their roles in Tau pathology. Using transmission electron microscopy (TEM), cryo-electron microscopy (cryo-EM), and a range of optical microscopy techniques, we discovered that these modifications generally hinder the in vitro assembly of Tau into PHFs. Interestingly, while acetylation's effect on Tau assembly displayed variability, either promoting or inhibiting phase transitions in the context of cofactor free aggregation, heparin-induced aggregation, and RNA-mediated liquid-liquid phase separation (LLPS), phosphorylation uniformly mitigated these processes. Our observations suggest that PTMs, particularly those situated outside the fibril's rigid core are pivotal in the nucleation of PHFs. Moreover, in scenarios involving heparin-induced aggregation leading to the formation of heterogeneous aggregates, most AD-specific PTMs, except for K311, appeared to decelerate the aggregation process. The impact of acetylation on RNA-induced LLPS was notably site-dependent, exhibiting both facilitative and inhibitory effects, whereas phosphorylation consistently reduced LLPS across all proteoforms examined. These insights underscore the complex interplay between site-specific PTMs and environmental factors in modulating Tau aggregation kinetics, enhancing our understanding of the molecular underpinnings of Tau pathology in AD and highlighting the critical role of PTMs located outside the ordered filament core in driving the self-assembly of Tau into PHF structures.
Organizational Affiliation:
Department of Chemistry, University of Colorado, Boulder, Colorado 80309, United States.