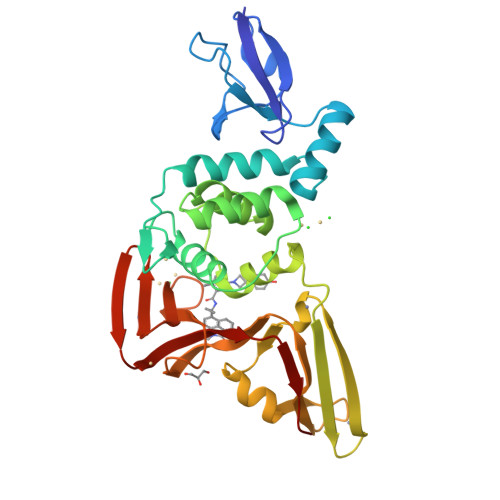
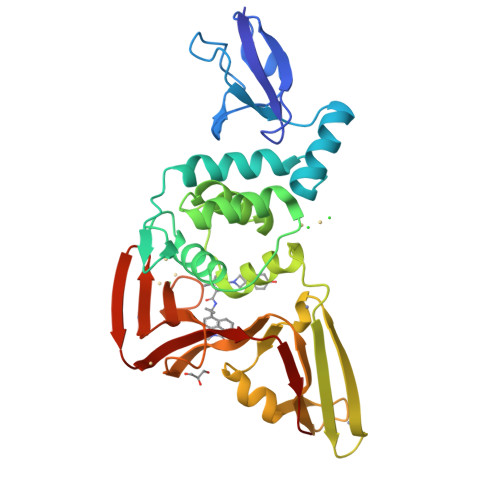
Discovery of orally bioavailable SARS-CoV-2 papain-like protease inhibitor as a potential treatment for COVID-19.
Lu, Y., Yang, Q., Ran, T., Zhang, G., Li, W., Zhou, P., Tang, J., Dai, M., Zhong, J., Chen, H., He, P., Zhou, A., Xue, B., Chen, J., Zhang, J., Yang, S., Wu, K., Wu, X., Tang, M., Zhang, W.K., Guo, D., Chen, X., Chen, H., Shang, J.(2024) Nat Commun 15: 10169-10169
- PubMed: 39580525
- DOI: https://doi.org/10.1038/s41467-024-54462-0
- Primary Citation of Related Structures:
8YX2, 8YX3, 8YX4, 8YX5 - PubMed Abstract:
The RNA-dependent RNA polymerase (RdRp), 3C-like protease (3CL pro ), and papain-like protease (PL pro ) are pivotal components in the viral life cycle of SARS-CoV-2, presenting as promising therapeutic targets. Currently, all FDA-approved antiviral drugs against SARS-CoV-2 are RdRp or 3CL pro inhibitors. However, the mutations causing drug resistance have been observed in RdRp and 3CL pro from SARS-CoV-2, which makes it necessary to develop antivirals with novel mechanisms. Through the application of a structure-based drug design (SBDD) approach, we discover a series of novel potent non-covalent PL pro inhibitors with remarkable in vitro potency and in vivo PK properties. The co-crystal structures of PL pro with lead compounds reveal that the residues D164 and Q269 around the S2 site are critical for improving the inhibitor's potency. The lead compound GZNL-P36 not only inhibits SARS-CoV-2 and its variants at the cellular level with EC 50 ranging from 58.2 nM to 306.2 nM, but also inhibits HCoV-NL63 and HCoV-229E with EC 50 of 81.6 nM and 2.66 μM, respectively. Oral administration of the GZNL-P36 results in significantly improved survival and notable reductions in lung viral loads and lesions in SARS-CoV-2 infection mouse model, consistent with RNA-seq data analysis. Our results indicate that PL pro inhibitors represent a promising SARS-CoV-2 therapy.
Organizational Affiliation:
Guangzhou National Laboratory, Guangzhou, 510005, China.