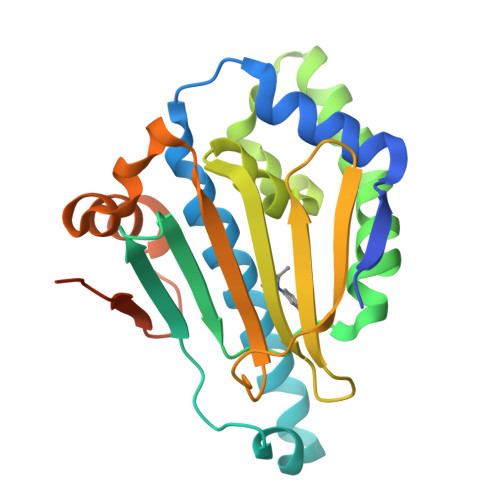
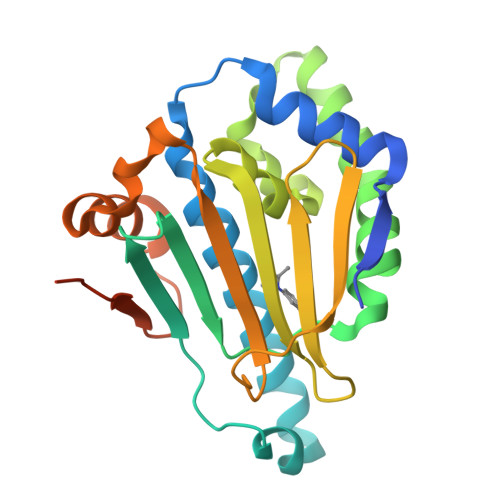
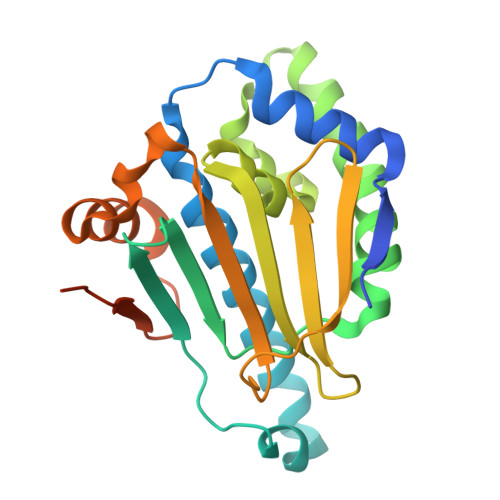
Novel starting points for fragment-based drug design against human heat-shock protein 90 identified using crystallographic fragment screening.
Huang, L., Wang, W., Zhu, Z., Li, Q., Li, M., Zhou, H., Xu, Q., Wen, W., Wang, Q., Yu, F.(2025) IUCrJ 12: 177-187
- PubMed: 39819741
- DOI: https://doi.org/10.1107/S2052252524012247
- Primary Citation of Related Structures:
7H9K, 7H9L, 7H9M, 7H9N, 7H9O, 7H9P, 7H9Q, 7H9R, 7H9S, 7H9T, 7H9U, 7H9V, 7H9W, 7H9X, 7H9Y, 7H9Z, 7HA0, 7HA1, 7HA2, 7HA3, 7HA4, 7HA5, 7HA6, 7HA7, 7HA8, 7HA9, 7HAA, 7HAB, 7HAC, 7HAD, 7HAE, 7HAF, 7HAG, 7HAH, 7HAI, 7HAJ, 7HAK, 7HAL, 7HAM, 7HAN, 7HAO, 7HAP, 7HAQ, 7HAR, 7HAS, 7HAT, 7HAU, 7HAV, 7HAW, 7HAX - PubMed Abstract:
Heat-shock protein 90 (HSP90) is a highly active molecular chaperone that plays a crucial role in cellular function. It facilitates the folding, assembly and stability of various oncogenic proteins, particularly kinases and transcription factors involved in regulating tumor growth and maintenance signaling pathways. Consequently, HSP90 inhibitors are being explored as drugs for cancer therapy. Crystallographic fragment screening is a novel screening method that has been developed in recent years for fragment-based drug discovery and is known for its high hit rate and its ability to provide direct insights into the complex structures of proteins and compounds. In this paper, high-diffraction-resolution crystals of the N-terminal domain of human HSP90α were employed in crystallographic fragment screening to discover binding fragments and binding sites. A diverse library of 800 structurally distinct fragments was screened, yielding 91 starting points for the fragment-based drug design of new HSP90α N-terminal inhibitors. Nearly a thousand crystals were measured, with 738 being processed and phased using a highly automated data-processing pipeline including data reduction and phasing, refinement and hit identification via PanDDA multi-data-set analysis. The 91 identified compounds bind to eight distinct regions of the HSP90α N-terminus, with 63 fragments located in the ATP-binding pocket and its surroundings, thus demonstrating the potential for the development of HSP90α- and ATP-binding inhibitors. This study emphasizes crystallographic fragment screening as a powerful method that can effectively identify fragment molecules and inhibitors that bind to HSP90α, contributing to ongoing efforts in cancer drug discovery.
Organizational Affiliation:
University of Chinese Academy of Sciences, Beijing 100049, People's Republic of China.